
Case Study:
Human Health Benefits, Operational Efficiencies, Increased Productivity, Cost Improvements
CHALLENGE
GlaxoSmithKline faced a waste bedding management challenge involving both laboratory animal allergies and ergonomic related employee injuries.
CUSTOMER
GlaxoSmithKline
Industry
Vivarium
GlaxoSmithKline (GSK) is a global company engaged in the research, development and manufacture of products ranging from prescription medicines, to vaccines, and consumer healthcare products. The company promotes its dedication to helping people live longer, healthier lives through research, new product development, global social responsibility initiatives, and disease prevention. Research is the cornerstone of GSK growth and supports its commitment to helping patients around the world. A small, but vital part of that work, involves animal research.
 GSK makes the care, welfare and treatment of its research animals a top priority. The GSK research animal program in Research Triangle Park (RTP), NC is accredited by the Association for the Assessment and Accreditation of Laboratory Animal Care International (AAALACI). This independent group reviews and assesses animal care programs against published standards and industry practices.
GSK makes the care, welfare and treatment of its research animals a top priority. The GSK research animal program in Research Triangle Park (RTP), NC is accredited by the Association for the Assessment and Accreditation of Laboratory Animal Care International (AAALACI). This independent group reviews and assesses animal care programs against published standards and industry practices.
Alongside the company’s commitment to the research animals is a likewise strong commitment to the health and well being of GSK employees. In the area of animal research, a strong focus has been built around protection of employees from ergonomic related injuries and laboratory animal allergies (LAA). In the early 1990s, GSK at RTP implemented its first respiratory protection program in the US for protection of employees against LAA.
In addition to the LAA implications, ergonomic related injuries can be quite common in a research animal program. GSK Environmental, Health and Safety (EHS) Laboratory Animal Science (LAS) teams have worked very diligently to understand where work practices can impact ergonomics. Both prevention of LAA exposure and ergonomic injuries were high on the list of goals in developing a new approach to management of animal waste bedding.
It is well established that exposure to animal proteins, via either inhalation or contact, can lead to the development of LAA. GSK EHS professionals have worked closely with the GSK LAS team to develop a more complete understanding of low, medium and high exposure and ergonomic risks associated with typical work scenarios. A hierarchy of protection is in place whereby engineering controls are promoted as the first defense against exposure and complimented by administrative, process and personal protection strategies. The monitoring and data collection by the EHS partners at GSK showed that the cage wash area contained the highest level of airborne particles experienced in the program, at >50 ng/m3, based on GSK exposure ratings.
This data, coupled with the correlation between the level and frequency of contact with the animal waste, requires the cage wash technicians to wear full personal protection equipment (PPE). These layers of draping, head cover, gloves and booties made the workers very uncomfortable and fatigued due to the hot and humid environment of the wash room.
THE CHALLENGES OF ANIMAL WASTE BEDDING MANAGEMENT
The GSK research animal facility involved in this project is a 124,611 sq ft cast-in-place concrete building spread over six levels, with four levels dedicated to animal housing and study support. This building is part of the main GSK research complex in RTP, N.C. With a primary population of rodents – mice and rats – the cleaning and maintenance of approximately 3,500 polycarbonate cages per week is a continuous process for the technicians assigned to the cage-wash operations.
 At the time of project initiation, LAS was running three wash areas in the building where the project was to be completed. Of the three, only one was truly fit for purpose based on size. Part of the overall strategy for process improvement, in addition to the new waste bedding system, was to consolidate wash area operations for the building into the largest wash room on level one of that building. This conclusion was reached through a technician led process improvement team and endorsed by LAS leadership.
At the time of project initiation, LAS was running three wash areas in the building where the project was to be completed. Of the three, only one was truly fit for purpose based on size. Part of the overall strategy for process improvement, in addition to the new waste bedding system, was to consolidate wash area operations for the building into the largest wash room on level one of that building. This conclusion was reached through a technician led process improvement team and endorsed by LAS leadership.
Prior to the waste management system upgrade, the waste bedding handling process consisted of several ergonomically inefficient steps, as well as exposure potential related to LAA. In the wash area, cages were lifted from the carts, upended to dump contents and often manually scraped to remove any clinging residue. Due to the location of the dump station in relation to the tunnel washer, technicians had to stoop, twist and rotate to move between the carts, waste bedding dump station and load end of the washer. At the final capture end of the waste system, technicians again had to be gowned up, manipulate heavy liners bags and tilt trucks capturing the soiled waste. Support personnel had to then transport the tilt trucks down hallways to a waste consolidation area for packaging suitable for over-road transport. Additional details about the process are explained later in this article.
The original pneumatic driven waste system, which was more of a waste collection system than a waste management system, was not compatible with bedding and enrichment products presently being used in the program. This resulted in frequent clogs to the system and overloading of the dust collection filters. When that system malfunctioned, technicians had to dump cage contents into waste boxes creating an additional LAA exposure and ergonomic impact. In addition, the GSK maintenance team would have to open the system for cleaning and repair. Every time repairs were made, more people were exposed to the dirty bedding and the allergens. The extensive down time also increased operating costs and decreased operating efficiencies.
Another disadvantage of the pneumatic process involved the waste separation end of the system. An entire room, located in a centralized area within the facility, had to be devoted to this process and occupied a place that could have been used for much needed equipment and supply storage. The airstream with the entrained bedding waste traveled through pipes to cyclone separators in the dedicated room where the separated waste was dumped into bag-lined tilt trucks. The extraction system was a closed system during most of the process but when the carts were changed, the dust and bedding particles generated by the activity in that room created an unacceptably high exposure risk. In addition to the exposure issues, when the weight limit for each tilt truck was reached, the system would alarm until one of the technicians went to the room, replaced the cart and re-set the system. This process took the technicians away from other duties and also posed potential ergonomic issues since the staff had to move these trucks out of the room to a loading dock for transport, move new trucks into place, and manage heavy-duty plastic bags that had to be inserted in the trucks. Other troublesome factors were the sheer weight of the bags on a roll, and the cost of the specially sized bags at $10,000 for a 100 bag roll.
In 2012, GSK closed their RTP on-site incinerator operations. With on-site incineration, the tilt trucks containing the bagged waste could be loaded onto a truck and, upon arriving at the incinerator, be directly dumped onto the incinerator floor. With closure of incinerator operation, the bagged waste had to be packaged for longer, over-the-road transport. The choice at that point was to make extensive modifications to the separation system or create a system to transfer filled bags to 1 yd3 shipping boxes. Because each bag of waste weighed around 200 lbs, a lift system had to be installed to pick the bags up out of the tilt trucks and deposit them in the box. With the long standing issues with pneumatic collection, GSK chose to pursue an alternative system which could be specifically designed to accommodate over-the-road transport.
A BETTER WAY TO MANAGE ANIMAL BEDDING
The GSK project team with members from LAS and GSK Capital Project Engineering partnered to analyze elements of the existing system that were problematic. They sought to identify which parts of the process would need to be improved by a new waste management system. Long-term experience of the team members in research animal facility operations provided a knowledgeable foundation upon which to build a new design. The team knew they had sound justification for proposing a project to replace the existing pneumatic system. Key components of the justification included:
- Insufficient engineering controls for prevention of LAA and ergonomic problems
- The amount and value of animal support space that was being consumed by the mechanical components supporting a pneumatic vacuum system
- The lack of dependability and inefficiencies with the existing system
- The environmental impact and cost related to the energy required to operate the pneumatic system
Taking input from technical specialists and utilizing the team’s extensive experience in laboratory animal management, engineering and operations, the team concluded that the supplier’s expertise must be in the field of material handling. Previous experience in research animal facilities and with the handling of waste bedding was desired but the supplier did not need to be associated with a supplier of other lab animal management systems. The team also insisted that the new system must:
- Utilize proven engineering practice to mitigate LAA exposure to workers while reducing the ergonomic impact of the waste management process
- Be designed for maintenance access and utilize proven material handling concepts to ensure consistent and reliable operation
- Operate with a minimum of noise and vibration outside of the cage wash area and not add operating noise that would increase the overall sound level above 80 dB
- Control airborne dust, associated with cage dumping, and have HEPA filtration prior to entering any mechanical equipment or leaving the wash room
- Be able to move waste mechanically from the cage wash area directly to the outside container
- Allow for waste to be sufficiently packaged at the end of the process to allow acceptance at commercial incinerator
- Be sealed from end to end to eliminate any handling of exposure to bedding after it was discharged into the dump hopper
- Include geographically close supplier support to quickly respond to any operational issues with the system beyond the capabilities of in-house personnel
As the development of the comprehensive scope progressed, the GSK team enlarged to include other partners in the design and implementation phase. ACH Constructors, LLC. is a local general contractor that has partnered with GSK on a wide variety of projects and Jacobs Engineering, a global A/E who has partnered with GSK for architectural and engineering services. Both companies have a long and successful history with GSK and significant expertise in pharmaceutical laboratory design, renovation and construction. Working with ACH and Jacobs, the GSK team began to research their options for a new waste bedding handling system.
Animal waste bedding can have many varying profiles depending not only on the type of bedding but other factors such as the particular therapeutic area of research, unique diets that end up in the waste stream, and the amount/type of animal waste being generated. Understanding the dynamics of material flow, the variability of material size and bulk density, the abrasive nature of the material, the optimization of the conveyor layout, the physical limitations of the facility to conveyor design, and the load out parameters as provided by the waste hauler are all key elements in determining the most effective waste bedding handling system for a research animal facility.
Several potential companies and products were considered and invited to submit proposals. One of those companies was Hapman. Through industry recommendations and discussions with Hapman, GSK concluded that Hapman would offer the material handling expertise that was needed to optimize the waste bedding project. It was obvious from the start that the Hapman team would bring rich, material handling expertise that was much needed as well as past experience with animal bedding. After much discussion and evaluation, the GSK team concluded that the Tubular drag chain conveyance system offered by Hapman would be the best option for handling the multi-faceted components of animal waste bedding.
POINT A TO POINT B: NO STRAIGHT LINE
The preliminary site visit and walk-through gave Hapman a good understanding of the layout and performance and design challenges for the new system including configuring the waste dumping stations, level changes, turns, bends, tight spaces, location restrictions and final waste capture. Hapman engineers worked with the GSK/ACH team to incorporate the collective knowledge of material handling, lab animal operations, facility requirements and construction to develop the final plan for the new system. The weekly planning meetings were the focal point for a review of the design as it was developed by Hapman. The design concepts and GSK requirements were openly and honestly challenged and discussed resulting in a robust give-and-take that helped everyone to remain engaged and vested in the design and construction phase.
As important as it was to control LAA exposure with engineered systems, the team did not lose sight of the impact that the ergonomics of the work flow had on the technicians. To help design the best system possible with the least amount of conflicting movements, Hapman provided a constant stream of design details and ACH came up with a unique solution to fully understand the technicians’ movements as they emptied cages. ACH used drawings of the cage dump station provided by Hapman and built a prototype of the equipment out of card board, PVC pipe and duct tape.
 |
 |
A cardboard mock up of the dump station was created to assist technicians in analyzing their movements and help the Hapman team customize a system that was more ergonomic, faster, and efficient.
The full-scale equipment models gave the technicians the ability to analyze their own movements during the cage dump process and provide immediate feedback to the team. The technicians became immediately engaged and moved the model around the wash room to find the best location that required the least twisting and bending. When they put the model cage dump station directly in front of the tunnel washer and discussed modification to the exhaust system, they knew they had found their answer.
the technicians the ability to analyze their own movements during the cage dump process and provide immediate feedback to the team. The technicians became immediately engaged and moved the model around the wash room to find the best location that required the least twisting and bending. When they put the model cage dump station directly in front of the tunnel washer and discussed modification to the exhaust system, they knew they had found their answer.
Putting the cage dump station in front of the tunnel washer eliminated a 90 degree turn and several additional arm and rotation movements from the process of taking the cages from the carts holding dirty cages, emptying them into the dump station and preparing them to enter the washer. The height of the dump station matched the height of the inlet conveyor for the tunnel washer, which meant less reaching and bending for the technicians. With the time savings these improvements brought to the cage dump process, the technicians could quickly and easily empty the carts of their soiled cages.
As previously mentioned, part of the strategy for process improvement alongside the main project was to consolidate all wash operations into one primary wash room on level one of the building. The desire was to centralize in order to take advantage of the best fit for purpose wash area in the building while retaining one other wash area for back-up. The remaining wash area would be closed down.
In light of this, the next design element was to minimize the conveying equipment necessary to take animal bedding from the reserve wash area to the main conveyor. The design challenge for the waste bedding system was to move material a distance of 250’ while navigating the existing mechanical space above the ceiling, and maintaining the existing drop points on each floor. When the Hapman design team examined the space above the drop ceiling, it was apparent there was no clear path to reach the destination. At this critical design point, GSK’s in-house maintenance partner, Johnson Controls, Inc. (JCI), was brought into the project team. Working closely with JCI and ACH, Hapman engineers were able to build a more complete understanding of the available interstitial space and how to accommodate the conveyor. Hapman then proceeded to design a custom Wye configuration that could make the transition from the 75’ Tubular Drag Conveyor on the first floor and the 30’ drop from the third floor into a single Tubular Drag Conveyor that would carry the material the last 115’ to a briquetter.
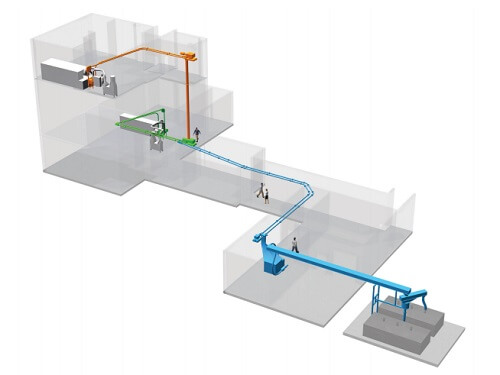
This custom Wye design kept the overall foot print small and eliminated several transitions that would have taken considerable space and the team’s decision to go with a drop eliminated a vertical conveying section and an additional drive from the system. The complete waste bedding handling system consisted of:
- A. The Tubular Drag Conveyor carrying the waste bedding 60’ from the reserve cage wash room, to a drop point
- B. A 30’ stand pipe that dropped waste bedding material from the third flue into the transfer conveyor
- C. The primary wash room Tubular Drag Conveyor to carrying the waste bedding 75 feet to the transfer conveyor
- D. In both wash rooms, a cage dump station, mounted directly in front of the tunnel washer, provided an optimal ergonomic cage dump process. Dust control for these stations was provided with a stand-alone HEPA air filtration system
- E. The custom engineered Wye piping configuration where the third floor stand pipe, and first and third floor Tubular Drag conveyors join to feed the main transfer conveyor
- F. The transfer Tubular Drag Conveyor that runs 115’ above the ceiling tiles down the long corridor, to the briquetter (discussed later in this article)
- G. The outlet of the Tubular Drag Conveyor that feeds the briquetter. The briquetter uses hydraulic shafts to create hockey-puck sized briquettes out of the bedding material
- H. The 45’ belt conveyor that carries the briquetted waste bedding out to the container
- I. The engineered load-out system with rotating conveyor: a 6’ swivel conveyor feeds the custom designed container which has three inlet ports for even filling of the container. The automated swivel conveyor allows a single operator to use a hand control to move the position of the loading tube to one of the three container inlets. In addition, the load-out system has the ability to swivel the conveyor to a second container when the first is full. Overall, the swivel conveyor can move through a 270º arc
- J. The briquetting system was purchased from a briquette equipment supplier by GSK and integrated into the conveying system by Hapman and ACH
 Once these design hurdles were solved, the project team focused their attention on the collection of waste at the end point of the waste stream. Because GSK has the ultimate goal of “zero to landfill” and a focus on the reduction of the use of natural resources, the project needed a commercial incinerator for final disposal of the animal waste bedding. The waste could not simply be collected and hauled away to a landfill. After the complexity of managing loose, waste bedding through a system of tilt trucks, bagging, lifting and loading into boxes, the LAS operations team was eager for a more streamlined approach.
Once these design hurdles were solved, the project team focused their attention on the collection of waste at the end point of the waste stream. Because GSK has the ultimate goal of “zero to landfill” and a focus on the reduction of the use of natural resources, the project needed a commercial incinerator for final disposal of the animal waste bedding. The waste could not simply be collected and hauled away to a landfill. After the complexity of managing loose, waste bedding through a system of tilt trucks, bagging, lifting and loading into boxes, the LAS operations team was eager for a more streamlined approach.
A key part of GSK’s focus on the minimization of exposure to LAA is that it applies to any workers, whether inside of GSK or to their external partners. With the assistance of GSK EHS personnel and their waste management partner, conversations were started with various incinerator operations. The initial concept for the final packaging of the waste was the use of a bulk bag container. The team was not fully satisfied with this direction but didn’t have a better idea. With the luck of serendipity, one of the Hapman project partners, Industrial Air, happened to wonder aloud if briquetting the waste might be an option. The GSK team was intrigued and pursued further information to support the idea.
 Ultimately, the GSK project team decided that briquetting would further support their desire to minimize exposure to LAA through limiting personnel contact with the waste bedding. Once again, the project team began to research possible partners for this leg of the project. After thorough review of possible vendors and request for proposals, Weima was selected as the partner for the briquetting component. Weima worked closely with the project team to incorporate a briquetter. The design, with interfaced final drag chain conveyor, transported loose waste bedding directly to the briquetter and then to an additional sealed conveyor that moved the briquettes from the briquetter to a sealed, external dumpster system. This design resulted in a single point of contact, at the dump station, for exposure by those handling the waste. The incinerator operation, due to their own personnel protection requirements, was already set up to allow for the briquettes to be dumped directly on the floor of their special waste area. Because the briquettes were densely compacted, LAA laden dust (typical to loose bedding) was no longer an issue. The briquettes offered additional value to GSK because that material takes up less space in the container and reduces the number of trips to the incinerator.
Ultimately, the GSK project team decided that briquetting would further support their desire to minimize exposure to LAA through limiting personnel contact with the waste bedding. Once again, the project team began to research possible partners for this leg of the project. After thorough review of possible vendors and request for proposals, Weima was selected as the partner for the briquetting component. Weima worked closely with the project team to incorporate a briquetter. The design, with interfaced final drag chain conveyor, transported loose waste bedding directly to the briquetter and then to an additional sealed conveyor that moved the briquettes from the briquetter to a sealed, external dumpster system. This design resulted in a single point of contact, at the dump station, for exposure by those handling the waste. The incinerator operation, due to their own personnel protection requirements, was already set up to allow for the briquettes to be dumped directly on the floor of their special waste area. Because the briquettes were densely compacted, LAA laden dust (typical to loose bedding) was no longer an issue. The briquettes offered additional value to GSK because that material takes up less space in the container and reduces the number of trips to the incinerator.
EXPERIENCE AND TEAMWORK: A MODEL FOR SUCCESS
After the detailed design process was complete and the approval of the Hapman waste bedding handling system was finalized, the team came together to establish a plan for implementation.
 For many companies, the association with the equipment manufacturer ends after the equipment leaves their facility. The user of the equipment is left with the installer to understand the technical installation requirements and enable a smooth start-up. Hapman, as part of standard practice, worked hand-in-hand with GSK to ensure the system would operate as specified. This meant Hapman provided a turn-key package to GSK, designing the entire bedding handling system, and all electrical and controls. The Hapman electrical team established the custom controls for normal operation, DCS monitoring from the GSK central system, and tie-in with the central alarm system. Hapman was responsible for the installation of the system, the start-up commissioning, and the site acceptance testing (SAT). The day-to-day oversight of these activities was managed by ACH who also took on the responsibility for the delicate process of ensuring that the installation process did not impact any ongoing studies.
For many companies, the association with the equipment manufacturer ends after the equipment leaves their facility. The user of the equipment is left with the installer to understand the technical installation requirements and enable a smooth start-up. Hapman, as part of standard practice, worked hand-in-hand with GSK to ensure the system would operate as specified. This meant Hapman provided a turn-key package to GSK, designing the entire bedding handling system, and all electrical and controls. The Hapman electrical team established the custom controls for normal operation, DCS monitoring from the GSK central system, and tie-in with the central alarm system. Hapman was responsible for the installation of the system, the start-up commissioning, and the site acceptance testing (SAT). The day-to-day oversight of these activities was managed by ACH who also took on the responsibility for the delicate process of ensuring that the installation process did not impact any ongoing studies.
The work schedule for the construction phase of the project was equivalent to the coordination, timing, and precision of a symphony orchestra. The research being performed at GSK at the time of construction required that the animals be unaffected by external noise and unnatural stress. There were some days when o
